Metamorphism in TDP-43 prion-like domain determines chaperone recognition
- Jaime Carrasco
- 1 mar 2023
- 3 min de lectura
Actualizado: 31 mar
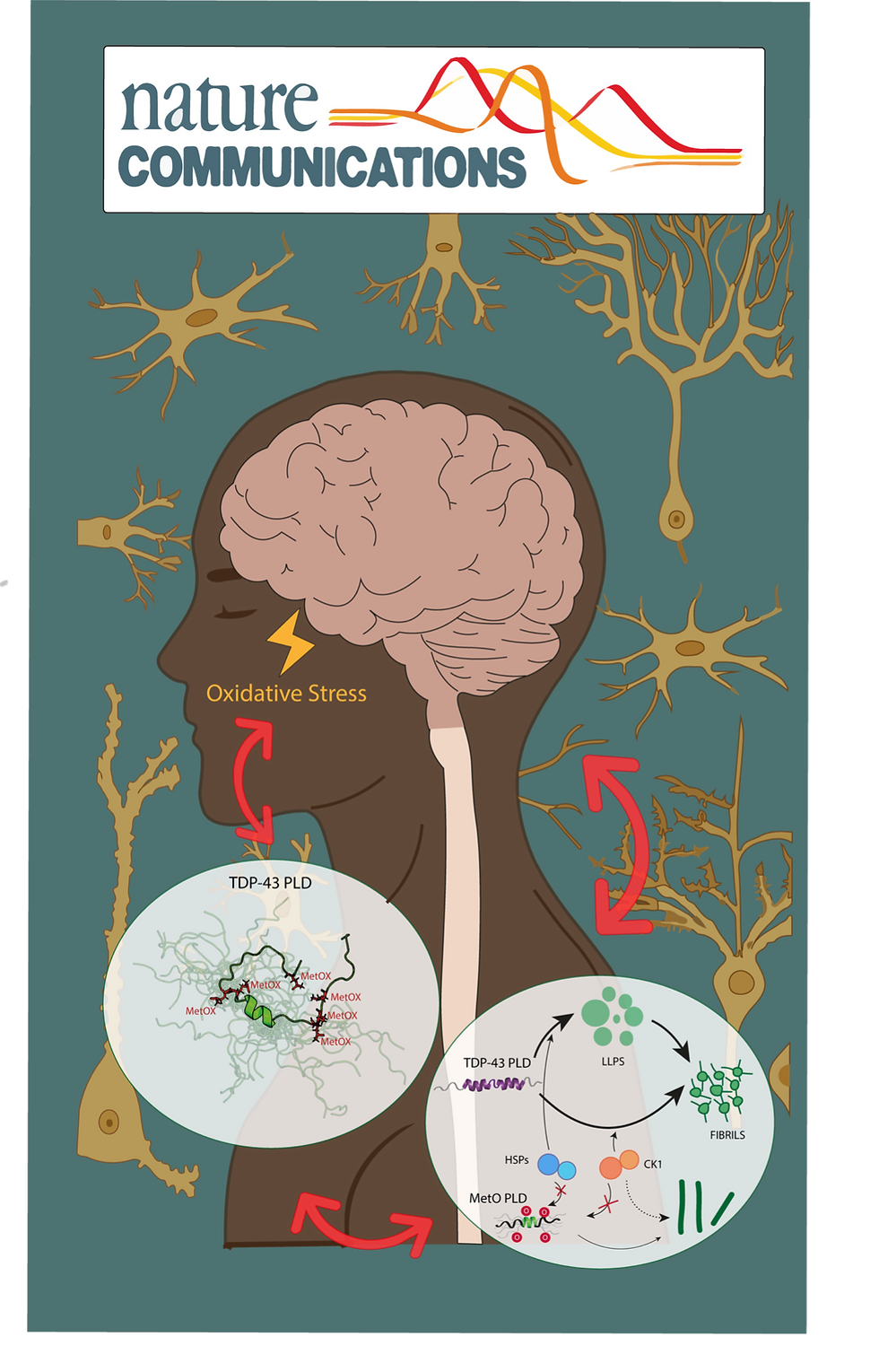
The RNA binding protein TDP-43 forms cytoplasmic inclusions via its C-terminal prion-like domain in several neurodegenerative diseases. Aberrant TDP-43 aggregation arises upon phase de-mixing and transitions from liquid to solid states, following still unknown structural conversions which are primed by oxidative stress and chaperone inhibition. Despite the well-established protective roles for molecular chaperones against protein aggregation pathologies, knowledge on the determinants of chaperone recognition in disease-related prions is scarce. Here we show that chaperones and co-chaperones primarily recognize the structured elements in TDP-43´s prion-like domain. Significantly, while HSP70 and HSP90 chaperones promote TDP-43 phase separation, co-chaperones from the three classes of the large human HSP40 family (namely DNAJA2, DNAJB1, DNAJB4 and DNAJC7) show strikingly different effects on TDP-43 de-mixing. Dismantling of the second helical element in TDP-43 prion-like domain by methionine sulfoxidation impacts phase separation and amyloid formation, abrogates chaperone recognition and alters phosphorylation by casein kinase-1δ. Our results show that metamorphism in the post-translationally modified TDP-43 prion-like domain encodes determinants that command mechanisms with major relevance in disease.
The Shape-Shifter Within: How TDP-43’s Structural Flexibility Dictates Neurodegeneration
In the complex machinery of the human brain, few proteins are as notorious as TDP-43. Known primarily for its role in RNA binding, TDP-43 has a darker side: it is the primary component of the protein inclusions found in patients with Amyotrophic Lateral Sclerosis (ALS) and Frontotemporal Lobar Degeneration (FTLD). But what exactly causes this protein to transition from a functional liquid state into toxic, solid aggregates?
A groundbreaking study published in *Nature Communications* (Carrasco et al., 2023) sheds new light on this mystery, revealing that the "shape-shifting" nature—or metamorphism—of TDP-43’s C-terminal prion-like domain is the key to how our cellular defense systems recognize it.
The Chaperone Connection
Cells employ a sophisticated "quality control" system known as molecular chaperones to keep proteins in check. Chaperones like HSP70 and HSP90 are the brain's first responders, preventing proteins from misfolding and clumping together.
The researchers discovered that these chaperones, along with various co-chaperones (specifically the HSP40 family), don't just randomly bind to TDP-43. Instead, they specifically target structured elements within its prion-like domain. Interestingly, while the main chaperones (HSP70 and HSP90) actually promote the liquid-liquid phase separation (LLPS) of TDP-43, different co-chaperones (like DNAJB1 and DNAJC7) have drastically different effects on how the protein "de-mixes."
The Oxidative Switch
One of the most significant findings of the study involves methionine sulfoxidation a chemical modification often triggered by oxidative stress.
The team found that when a specific helical element in the TDP-43 prion-like domain is dismantled by this modification:
- Phase separation and amyloid formation are significantly impacted.
- Chaperone recognition is abrogated, meaning the cell's defense system can no longer "see" or manage the protein effectively.
- Phosphorylation patterns are altered, further contributing to the disease-associated state of the protein.
Why It Matters
This "metamorphism¨ the ability of the TDP-43 domain to switch between different structural states—acts as a regulatory code. By understanding how oxidative stress and post-translational modifications change this code, scientists can better understand the "priming" events that lead to neurodegenerative disease.
This study doesn't just explain how TDP-43 aggregates; it identifies the specific structural "handles" that chaperones use to keep the protein healthy. In the long run, this could lead to new therapeutic strategies aimed at stabilizing these structures or enhancing chaperone recognition to prevent the onset of ALS and other devastating conditions.
Reference:
Carrasco, J., Antón, R., Valbuena, A. *et al.* Metamorphism in TDP-43 prion-like domain determines chaperone recognition. *Nat Commun* **14**, 466 (2023). [https://doi.org/10.1038/s41467-023-36023-z](https://doi.org/10.1038/s41467-023-36023-z)


Comentarios